 For example, if there are three p orbitals, five d orbitals, and seven f orbitals, electron pairing begins with the p, d, and f orbitals with the fourth, sixth, and eighth electrons. Hund’s rule asserts that electron pairing in orbitals belonging to the same subshell does not occur until each orbital in that subshell possesses one electron. The two portions of Hund’s rule describe how to fill a subshell containing many orbitals. Many subshells, however, include more than one orbital. The Pauli exclusion principle describes how electrons occupy a single orbital. 
Hund’s Rule or Hund’s Rule of Maximum Multiplicity This rule can also be used to describe a periodic table.It aids in demonstrating how the elements work together to generate chemical bonds.This certain principle can be used to explain a wide range of physical events.Significance of Pauli’s Exclusion Principle: If we construct a diagram of a helium atom, we can see that it has one electron up and one electron down. There are two electrons in the 1s subshell: n=1,l=0,ml=0. The atom is connected to two electrons, which occupy the outermost shell with opposing signs. Furthermore, unlike fermions, bosons can share or have the same quantum statesĪs an example of Pauli’s Exclusion principle, consider a neutral helium atom. It is unimportant for particles with integer spins, such as bosons, which have symmetric wave functions. Wolfgang Pauli developed the principle in 1925. Pauli’s Exclusion principle applies not just to electrons but also to other particles such as half-integer spin. Two electrons in the same orbital have opposing spins or are antiparallel.Pauli’s Exclusion principle has two rules: Only two electrons can coexist in the same orbital, and they must have opposing spins. No two electrons in an atom may have the same set of four quantum numbers, according to Pauli’s Exclusion principle. Pauli’s Exclusion Principle essentially helps us comprehend the electron configurations in atoms and molecules and also provides an explanation for the periodic table’s classification of elements. It is critical for students to understand, especially when studying electrons. In chemistry, Pauli’s Exclusion Principle, along with Aufbau’s Principle and Hund’s Rule, is one of the most significant principles. The spin angular momentum associated with electron spin is distinct from the orbital angular momentum associated with electrons traveling around the nucleus. Spin, like charge and rest mass, is a fundamental, unchanging feature of the electron. This angular momentum’s magnitude value is fixed. The quantum property of electrons is electron spin. Every shell is made up of one or more subshells, which are made up of atomic orbitals and are referred to as subshells. The electron shells are denoted by the letters K, L, M, N, O, P, and Q, or by the numbers 1, 2, 3, 4, 5, 6, and 7, counting from the innermost to the outermost shell. 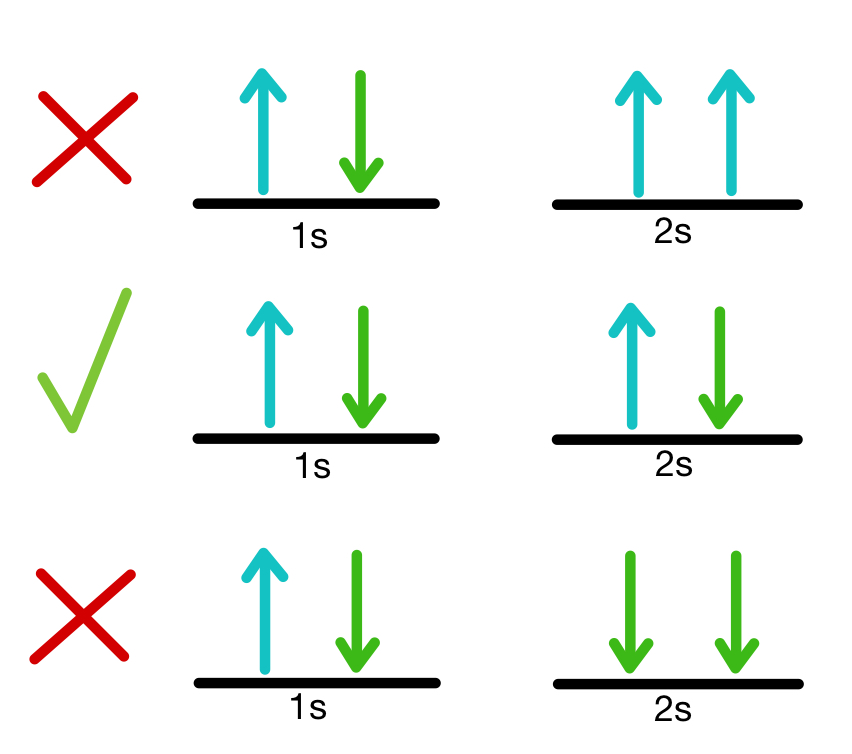
Electrons are supplied in such a way that a high constant configuration is achieved. An atom is made up of subatomic particles such as electrons, protons, and neutrons, with only the number of electrons being taken into account for electronic arrangement. It is the method or distribution of electrons in an atom’s orbitals. Chemists can anticipate an atom’s attributes, such as stability, boiling temperature, and conductivity, using the electron configuration and physical principles. The electron configuration of an atom is the orbital description of the electron locations in a typical atom. Electrons are much smaller than protons and neutrons, weighing over 1,800 times less than either. In quantum mechanics, the Pauli exclusion principle states that Fermions must have an antisymmetric wave function, unlike Bosons, which have symmetric wavefunctions. Hund’s rule uses the Pauli principle to fill up electrons in the various energy sublevels, thereby building up the periodic table elements.īesides, this principle applies to quantum mechanics and astrophysics. It can explain how atoms form chemical bonds and how chemical properties vary among the elements. Pauli exclusion principle can explain the electron-shell structure of atoms and the electron configuration of elements. 
Pauli principle also applies to atoms with half-integer spin. On the other hand, it does not apply to Bosons that have integer-spin (S = 1,2,3…). These particles do not violate the principle. Pauli exclusion principle applies to a set of fundamental particles called Fermions that have half-integer spin (S = 1/2, 3/2, 5/2 …). 
Pauli Exclusion Principle Example Applications 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
